DSHEA and the Profit-Driven World of Supplements: What Consumers Need to Know
The dietary supplement aisle is a $50+ billion industry in the United States alone. Rows of colorful bottles promise stronger immunity, sharper focus, better sleep, and ageless vitality. The labels sparkle with buzzwords—natural, detoxifying, clinically proven—but behind the glossy packaging lies a story of profit, politics, and loose regulation. To understand how supplements are marketed and sold today, we have to rewind to 1994, when a single law forever changed the supplement industry: the Dietary Supplement Health and Education Act (DSHEA).
How We Got Here: The Road to DSHEA
Before DSHEA, supplements were regulated much like food additives or drugs. Companies had to prove safety, justify claims, and sometimes even submit evidence to the FDA before going to market. The supplement industry argued these requirements were stifling innovation, preventing consumer access to “natural health options.”
The debate wasn’t only about regulation—it was also about fear. Industry-funded campaigns warned the public that the government might “take their vitamins away.” Ads featured everyday Americans holding up pill bottles with slogans like, “Don’t let the FDA take your health freedom.” The message worked. Citizens wrote to Congress, signed petitions, and demanded looser rules.
Enter DSHEA. Passed in 1994, it was championed by powerful lawmakers like Senator Orrin Hatch of Utah, whose state was—and still is—a hub for supplement manufacturing. The law gave the supplement industry the freedom it wanted, but at a cost: consumer protection and scientific rigor.
What DSHEA Actually Did
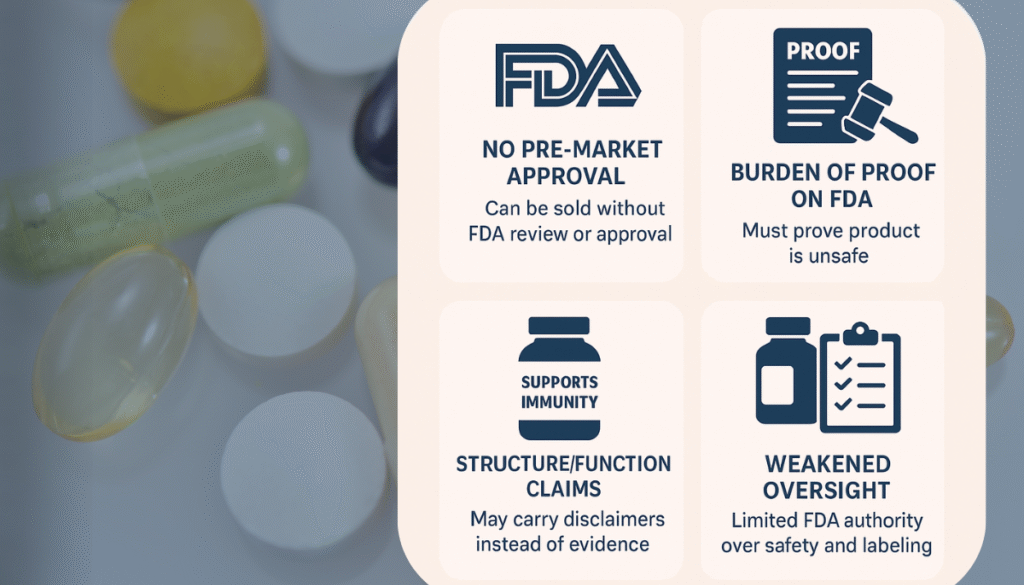
DSHEA shifted the regulatory balance in favor of industry, not consumers. Here’s what changed:
-
No Pre-Market Approval: Unlike drugs, supplements no longer required FDA approval before hitting the shelves. Companies can sell products without proving they’re safe or effective.
-
Burden of Proof on the FDA: Instead of manufacturers proving safety, the FDA must prove a supplement is unsafe to remove it from the market—often only after harm has already occurred.
-
Structure/Function Claims Allowed: Companies can say a product “supports immunity” or “boosts energy” as long as they include a tiny disclaimer: “This statement has not been evaluated by the FDA. This product is not intended to diagnose, treat, cure, or prevent any disease.”
-
Weakened Oversight: Manufacturers do not need to share test results or manufacturing data with the FDA unless specifically asked.
In short, DSHEA handed the keys of the industry to the companies making the products while stripping the FDA of much of its power to act proactively.
The Rise of a Profit-Driven Industry
With minimal barriers to entry, the supplement market exploded. Entrepreneurs and big corporations alike saw supplements as low-risk, high-reward ventures. The combination of DSHEA’s lax regulations and Americans’ hunger for natural health solutions created the perfect storm.
Marketing quickly outpaced science. Companies leaned into emotional advertising:
-
“Detox teas” promised to flush out mysterious “toxins.”
-
Memory boosters preyed on fears of dementia.
-
Immune boosters surged every cold-and-flu season.
Instead of investing heavily in clinical trials, companies invested in packaging, celebrity endorsements, and social media influencers. The result? Billions in profits, but little assurance for consumers about whether products actually work.
DSHEA’s Legacy: When Marketing is Louder than Medicine
Imagine walking into a car dealership where none of the vehicles had been safety-tested, but each one came with glossy brochures promising “ultimate performance” or “the car your family can trust.” That’s essentially what the supplement aisle looks like today.
DSHEA’s framework allows supplements to be marketed on suggestion rather than scientific proof. If a company can word its claims carefully enough—say, “supports mood” instead of “treats depression”—it can bypass stricter drug-level scrutiny.
Why This Matters for Consumers
The implications are serious:
-
Unrealistic Claims: Many supplements rely on buzzwords like “miracle cure” or “ancient secret,” which carry no scientific weight.
-
Hidden Risks: Supplements can interact with medications, cause side effects, or be contaminated with heavy metals or prescription drugs.
-
Wasted Money: A pill may contain so little of the active ingredient that it’s biologically useless. Proprietary blends, often a mix of dozens of ingredients under one label, make it impossible for consumers to know what they’re actually getting.
-
False Security: Believing that a supplement can “cure” a serious condition may delay someone from seeking real medical treatment.
Real-Life Examples of DSHEA’s Consequences
-
Ephedra (1990s–2000s): Marketed for weight loss and athletic performance, ephedra was linked to strokes, heart attacks, and deaths. It took years of mounting evidence before the FDA banned it in 2004.
-
DMAA Pre-Workout Supplements: Sold as a “natural stimulant,” DMAA was actually a synthetic amphetamine-like compound. It led to multiple deaths before finally being removed from the market.
-
CBD Boom: Largely unregulated, CBD supplements exploded after 2018. Products vary widely in quality and dose, with many containing far less (or more) CBD than advertised.
The Consumer’s Dilemma: Trust vs. Skepticism
Most supplement users are not reckless. They’re health-conscious people trying to fill nutritional gaps or optimize well-being. But DSHEA leaves them vulnerable to misinformation.
Think of it like online shopping from a website with no return policy, no reviews, and no guarantees—except you’re putting what you buy directly into your body. That’s the gamble consumers face every time they pick a supplement off the shelf.
What Consumers Can Do: A Practical Guide
Until laws change, the burden of responsibility largely falls on consumers. Here are some steps to protect yourself:
-
Look for Third-Party Certification: Seals from USP, NSF, or ConsumerLab indicate at least some level of independent testing.
-
Check Evidence, Not Hype: Search PubMed or NIH’s Office of Dietary Supplements for real research, not just marketing claims.
-
Beware of “Too Good to Be True” Claims: If a product promises to cure everything from cancer to acne, it’s a red flag.
-
Be Skeptical of Proprietary Blends: If the label doesn’t disclose exact amounts, you may not be getting an effective dose.
-
Talk to a Healthcare Professional: Pharmacists, physicians, and registered dietitians can help assess whether a supplement makes sense for your needs.
Looking Ahead: Could DSHEA Change?
Calls for reform are growing louder. Critics argue that DSHEA, written before the rise of e-commerce and global supply chains, is outdated. With today’s supplement industry more complex than ever, many experts believe it’s time for stricter oversight.
Yet, industry lobbyists remain powerful, and change will not come easily. For now, consumer education is the best defense.
Conclusion: Navigating a Market Built on Profit
The supplement industry, empowered by DSHEA, is a marketplace where profits often come before proof. This doesn’t mean all supplements are useless—many have genuine benefits supported by strong evidence. But the system as it stands forces consumers to act as their own regulators.
By staying informed, asking critical questions, and leaning on credible science rather than shiny labels, you can make choices that truly support your health.